 Case study iSpray – External lubrication system
Case study iSpray – External lubrication system
The Challenge Punch sticking, hardness stability, and high ejection…
 What is the cornerstone of flawless IPC testing?
What is the cornerstone of flawless IPC testing?
Advantages of In-process Quality Control Testing Atline In-process tablet…
 ATEX Certification
ATEX Certification
Pharma Technology just got certification following the ATEX Directive…
 PHARMA TECHNOLOGY reveals its growth strategy 2025
PHARMA TECHNOLOGY reveals its growth strategy 2025
Pharma Technology, with more than 25 years of experience…
 EXHIBITION | PCI Days - Warsaw, Poland - 8-9 September 2021
EXHIBITION | PCI Days - Warsaw, Poland - 8-9 September 2021
Meet us at PCI Days in Poland (September 8th &…
 New partner for Q-Control Division
New partner for Q-Control Division
We are delighted to enter into a new collaboration…

The new CW-150 checkweigher from Pharma Technology was launched in April 2021 and combines innovative inline solution with modern design, OEB 3 level and user-friendly GMP compliant software.
OPC UA communication protocol ensures rapid and safe data handling for reporting, as well as communication with upstream/downstream equipment.
The format changes are simple & fast thanks to an assisted HMI guidance.

Each individual capsule or tablet is measured at up to 150.000 product per hour with an accuracy of 1 mg (3σ), and the resulting mass values determine if the product is accepted or rejected.
Thus, the CW-150 can act as a final quality tool preventing all non-compliant products from reaching the market or as a Quality by Design (QbD) approach using mass values to monitor upstream process steps.
Application Fields
The CW-150 can be used in multiple applications as an off-line or in-line tool. Typical off-line applications are the proof of global batch quality and the use of a batch saver inspection machine if the batch did not pass the quality check. Subsequently, the batch can be supplied to the machine, the capsules/tablets are measured, and all non-conforming products are rejected.


The release quality checks can be repeated with the remaining product and the batch can be released.
For the in-line mode, the CW-150 allows 100% weighing of capsules or tablets and thereby shorten the reject strategy process compared to a sampling method.
The CW-150 is OEB 3 level approved and can be upgraded with Near Infrared technology to become a CU-120 inspection machine for each individual tablet inspection in terms of thickness, diameter/length mass and API fraction to obtain the Content Uniformity.
CW-150 Inspection Technology
The advanced 3D microwave resonator is the heart of the CW-150. One machine PLC controls three individual sets of generators and receivers for the 3 space directions x- y and z. First, three resonance curves are established in the cavity without any product defining the baseline. After that one tablet or capsule enters the cavity, three fields will be applied to the product and all three curves will be affected due to the presence of product in the cavity. The difference between the three resonance curves with and without product are frequency shift A and half width broadening curves difference vs. time B, an overall six values per product are measured. During the measurement, the product is exposed to the 1-Watt microwaves for a few milliseconds. The energy transfer onto the product is therefore very small and insignificant in terms of risk to the product and changes in molecular structure.

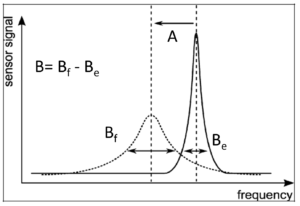
For calibration, the CW-150 uses a 7-dimensional regression based on the six values per product measured in the cavity and the mass is determined by rejecting these calibration products on the integrated high precision scale in the bottom of the CW-150. This calibration is a rolling calibration: in the inspection mode, some samples are directed to the scale measuring and comparing the predicted weights from the 3D microwave resonator with the real weights provided from the scale. If needed, the calibration will be applied using the new values.

For the use of tablets, the scale can be replaced by an integrated 4-Parameter tester measuring further physical attributes of the tablets such as mass, thickness, diameter/length, and hardness.

A detailed view on the Process of the CW-150
The products are fed into the feed hopper and the min. and max. product filling level is monitored by a capacity sensor. After reaching the vibrating chute through an opening in the bottom of the feed hopper, product fragments and/or excess of powder fall through a perforated plate integrated in the pre-feeder vibrating chute, while empty capsules, half capsules and poorly filled capsules are eliminated by compressed air during their journey through the vibrating chute.
From the vibrating chute, the products are transferred to the centrifugal bowl where the filling level is regulated by a laser sensor. The products exit the centrifugal bowl aligned and singularized, and continue their way guided, one by one, on a vacuumed conveyor.
Following this step, the products enter the 3D microwave resonator accelerated by compressed air injection, allowing only one product at a time to be in the cavity (measuring chamber) for mass measurement. All reference values for the rolling calibration loop are taken on the embedded weigh cell and the sample measurements are added to the general batch report of the current batch.
During production, mass values (trends and statistics) are displayed on the central HMI. Batch reports contain batch inspection statistics and histograms of the inspected population. “Good” products go straight into the product container, while a high-speed pneumatic system diverts “out of tolerance and out of plausibility” products to a rejection bin. Rejections are confirmed by a count sensor.
Conclusion
With the ground-breaking CW-150, Pharma Technology covers the high-speed inspection segment for pharmaceutical processes and provides an all-in-one solution for off-line, at-line and in-line applications. The main advantages of the CW-150 are: its multi-purpose applicability such as off-line and in-line measurements, Final Quality Inspection vs. Quality by Design approach, its ability to inspect all capsules and tablets under OEB 3 security before release to the market and the possibility to upgrade it into a CU-120 for additional Near Infrared analysis to obtain the Content Uniformity of each individual product.
CW-150 – Checkweigher [Mg] for tablets & capsules | Pharmatec